Environmental constraints on faunal patterns: an example in Early Ordovician (Arenig) trilobite assemblages from the Argentine Cordillera Oriental
Beatriz G. WAISFELD1 and Ricardo A. ASTINI1
1 Cátedra de Estratigrafía y Geológía Histórica, Facultad de Ciencias
Exactas, Físicas y Naturales, Universidad Nacional de Córdoba, Av. Velez
Sarsfield 299, 5000 Córdoba, Argentina; CONICET. E–mail: bwaisfeld@com.uncor.edu
Key words: Trilobites. Faunal patterns. Early Ordovician. Cordillera Oriental. Argentina.
Introduction
The Acoite Formation (basal Arenig – middle Arenig) yields
two contrasting lower Ordovician trilobite assemblages: the Thysanopyge
Fauna and the Famatinolithus Fauna. These assemblages display significant
differences in taxonomic composition, geographic distribution and diversity
patterns throughout the Cordillera Oriental. Hence, they represent an
interesting case study for analyzing major environmental controls on faunal
distribution, turnover, and replacement.
Most significant faunal events include (1) the radiation of the Thysanopyge Fauna with a high proportion of endemics, and (2) an important faunal turnover involving the extinction of major representatives of the Thysanopyge Fauna and the subsequent expansion of the Famatinolithus Fauna (local radiation + immigration of perigondwanan taxa) (cf., Waisfeld et al., 1999). Thus, our aim is to interpret the distribution of the Thysanopyge and Famatinolithus faunas across the shelf profile, to constrain environmental controls on diversity patterns, and to understand the significant turnover that takes place between both assemblages. The development of the Thysanopyge Fauna and the shift to the Famatinolithus Fauna is best recorded in the northern part of the Cordillera Oriental (Santa Victoria region), whereas the Famatinolithus Fauna is well–developed in the western region (Los Colorados–Chamarra region) thus the analysis will be focused in faunal and environmental changes documented in these areas (Figure 1).
Distribution and ecological features of the trilobite assemblages
The Thysanopyge Fauna (latest Tremadoc – lowermost middle Arenig), first defined by Harrington and Leanza (1957) is mainly composed of asaphids including several endemics (e.g., Thysanopyge, Araiocaris, Megalaspidella, Niobides, etc.); other trilobite families are subordinate in abundance (Figure 1). Arenig occurrences of this fauna are geographically restricted to eastern localities of the Cordillera Oriental. The Thysanopyge Fauna is mainly developed in nutrient–rich, scarcely bioturbated black shales, deposited in subtidal environments, below fair–weather wave base, under low oxygen conditions. It displays low within habitat diversity with low richness at generic and familial levels. Thysanopyge is represented by at least 5 different species (Waisfeld and Vaccari, in prep.) occurring at different localities and forming either low diversity assemblages (associated with one or two asaphid genera) or monospecific assemblages.
From an ecological viewpoint dominance of a single family
accounts for a relatively low ecological differentiation, including a limited
range of feeding strategies. Fortey and Owens (1999) proposed a predatory/scavenger
habit for the Ordovician trilobites of the family Asaphidae. In particular, the
large–sized Thysanopyge (maximum sagittal length of 20–25 cm) fits well
within this category. However, a broad range of disparity in size among the
asaphids of the Thysanopyge Fauna might account for habitat partitioning
and a further differentiation within the predatory/scavenger style (e.g sagittal
length of the other asaphids is no greater than 8 to 12 cm).
The Famatinolithus Fauna (late lower Arenig – middle Arenig) is considerably more diverse than the Thysanopyge Fauna, including trilobites belonging to several families apart from asaphids (Figure 1). These faunas share no taxa, except for the olenid Hypermecaspis. The Famatinolithus Fauna developed in high energy, well oxygenated, shallow environments from the inner shelf to the upper shoreface with variable bioturbation, wave, and tide influence. Higher diversity at familial and generic levels is also reflected in the broader array of trilobite morphotypes (Fortey and Owens, 1990) and a greater variety of feeding categories suggesting an increase in ecological complexity. According to the model for trilobite feeding habits proposed by Fortey and Owens (1999) predators/scavengers might have been represented not only by asaphids but also by calymenids (e.g., Neseuretus, Colpocoryphe), and pliomerids (e,g, Pliomeridius); filter feeders (with filter chambers) (e.g., Ampyx, Famatinolithus?) and particle feeders (e.g., Porterfieldia) might have also been present.
The Famatinolithus Fauna shows a clear gradient of diversity decline toward shallower waters (Figure 1). Some core taxa (e.g., Famatinolithus, Pliomeridius, Branisaspis) seem to be environmentally tolerant and widespread, persisting across the whole environmental range of the Famatinolithus Fauna, being joined by Ogyginus in the shallowest end of the bathymetric profile.
Environmental reconstruction
Facies analysis of the Early Ordovician successions exposed along the Cordillera Oriental has pointed out various characteristics common to deltaic environments (e.g., Astini and Waisfeld, 1993) and usually regarded as related to shoreline advance or retreat, without implying the depositional setting that provided the abundant sediment source to this largely shallow–marine setting developed for more than 1000 km N–S trend. The only existing quantitative model for the Ordovician stratigraphy of the Central Andean basin was provided by Bahlburg and Furlong (1996) and, although largely based on the Puna Basin record, it allows understanding the broad basin picture. In their model they suggested important arc related loading that triggered back–arc subsidence. This flexure phase would have helped to largely founder the Tremadoc platform, generating a relative sea–level rise that contributed to "restrict" the foreland. Around late Early Arenig time to mid Arenig (uppermost deflexus to lowermost bifidus Zones) deltaic influence on the deep largely dysareobic foreland shelf–ramp started. It is represented by a progressive transition from laminated black shales to non–laminated green shales implying better–oxygenated bottoms, increase in average grain size (silty beds), higher rates of both sedimentation and bioturbation, and more frequent event beds including parautochthonous and allochthonous thin bioclastic concentrations. It also allows inferring higher availability of nutrients and suspended fines. This transition is represented along the Cordillera Oriental with variable expressions as a result of both onshore–offshore across–shelf progradation, and along–shore variations due to lateral migration and shifting of deltaic sites or as a product of differential bypassing and drift of the different sedimentary loads. In the Santa Victoria area, close to the border with Bolivia, this transition also shows a strong faunal turnover. A strong facies change is also observed farther south along the western border of the Cordillera Oriental where Astini and Waisfeld (1993) documented progressive deltaic influence from the uppermost early Arenig in the middle–upper part of the Acoite Formation (Figure 1). This shift in facies is broadly coincident with the initial development of the Famatinolithus Fauna. Given how far up section this faunal assemblage is recorded, it is suggested that some members could have been able to tolerate water salinity variations due to the intimate association with significant tidal influence and intermittent subaerial exposure therein recorded. Deltaic conversions (Dalrymple et al., 1992) implying dominant–processes changes (e.g., wave–dominated to tide–dominated; subaerial exposure; deltaic to estuarine) do not seem to alter the Famatinolithus Fauna although they do seem to exert influence on diversity. Although the time constraint for the Thysanopyge Fauna and the Famatinolithus Fauna is relatively well known (about 5 Ma and 2 Ma respectively), it has to be kept in mind that their time resolution is much less than the scale at which the changes and dynamics of modern deltaic settings are understood. Most interesting results the velocity to which environmental shifts are recorded within deltaic settings, particularly when forced by external factors, the Mississippi delta Recent history being a good example. This allows ruling out autocyclic phenomena to explain the observed changes in the composition of these faunas.

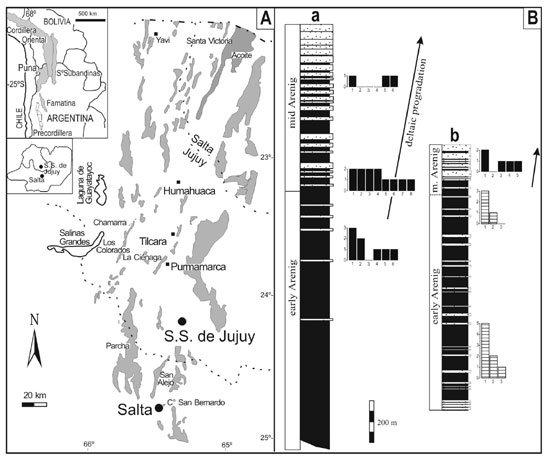
Figure 1. A. Location of Ordovician basins in southern South
America and outcrop map of the Santa Victoria Group. B. Simplified
columnar sections of the Acoite Formation in the western (a) and eastern (b)
parts of the Cordillera Oriental. Histograms show familial diversity of the
Thysanopyge (striped bars) and Famatinolithus (black bars) faunas in
selected portions of the onshore–offshore profile. Y axis:
number of trilobite genera within each family; X axis: trilobite families (1:
asaphids, 2: olenids, 3: calymenids, 4: raphiophorids, 5: trinuecleids, 6:
pliomerids, 7: nileids, 8: telephinids).
Conclusions
Arenig strata of the Cordillera Oriental record two
contrasting trilobite assemblages. A close match between environmental
peculiarities and their composition, diversity patterns and habitat distribution
is recognized. The Thysanopyge Fauna thrived in a relatively deep, low
energy, largely dysaerobic, restricted foreland shelf ramp. Environmental
conditions associated with this setting may have been responsible for its low
diversity and low ecological complexity. Foreland restriction
during the early Arenig was likely incidental in the relatively high endemism
displayed by the Thysanopyge Fauna. Arenig geographical restriction of
the Thysanopyge Fauna to the eastern Cordillera Oriental and its absence
farther west, in deeper offshore settings is presently unclear, however, subtle
changes in environmental/geochemical conditions might have been enough to
prevent its expansion.
The turnover from the Thysanopyge Fauna to the
Famatinolithus Fauna was triggered by the onshore–offshore progradation of
deltaic sites over the restricted, dysaerobic foreland shelf ramp. This change
produced the elimination of preferred habitats causing the extinction of the
Thysanopyge Fauna. The basin–wide expression of the Famatinolithus
Fauna (including several localities of the Bolivian Cordillera Oriental, cf.
Pribyl and Vanek, 1980; Waisfeld et al., 1999) accounts for the
widespread nature of the environmental and faunal change. Progradation of
deltaic sites resulted in higher–energy settings, changed circulation patterns
and brought well–oxygenated waters, with higher nutrient supply and frequent
changes in suspended fines. These new ecological settings, developed since the
uppermost lower Arenig to mid Arenig, controlled the turnover in taxonomic
composition and ecological properties, and favored the expansion of a broader
array of trilobite morphologies and feeding styles. The Famatinolithus
Fauna flourished through a relatively wide range of environmental possibilities,
even at the shallowest extreme of the environmental gradient. It successfully
adapted to heterogeneous habitats as a result either of onshore–offshore across–shelf
progradation or of along–shore variations, as well as to deltaic conversions,
only through changes in diversity (dominance of more eurytopic genera).
Accordingly, our results emphasize the effect of allocyclic changes in
the turnover of the assemblages, with a lesser role of authocyclic phenomena in
the configuration of these widespread trilobite faunas.
Acknowledgements
The authors thank financial support from CONICET and ANCyT–FONCYT through grants PICT 99 Nº 5387 and PICT 2000 Nº 8920.
References
Astini, R.A. and Waisfeld, B.G. 1993. Análisis estratigráfico y paleoambiental del Ordovícico medio (Formación Acoite y Sepulturas) en el borde occidental de la Cordillera Oriental jujeña. Actas 12º Congreso Geológico Argentino y 2° de Exploraciones de Hidrocarburos, Mendoza, 1: 96–106.
Bahlburg, H. and Furlong, K. 1996. Lithospheric modeling of the Ordovician foreland basin in the Puna– NW Argentina: On the influence of arc loading on foreland basin formation. Tectonophysics, 259: 245–258.
Dalrymple, R.W., Zaitlin, B.A. and Boyd, R. 1992. Estuarine facies models: conceptual basis and stratigraphic implications. Journal of Sedimentary Petrology, 62: 1130–1146.
Fortey, R.A. and Owens, R. 1990. Trilobites. In: Mc Namara, K.J. (ed.), Evolutionary trends, Belhaven Press, London: 121–142.
Fortey R.A. and Owens, R. 1999. Feeding habits in trilobites. Palaeontology, 42: 429–466.
Harrington, H., Leanza, A., 1957. Ordovician trilobites of Argentina. Department of Geology, University of Kansas, Special Publication 1, University of Kansas Press, Lawrence: 1–276.
Pribyl, A. and J. Vanek. 1980. Ordovician trilobites of Bolivia. Rozpravy Ceskoslovenské Akademie Ved. Rada Matematických a Prírodrích Ved, 90: 1–90.
Waisfeld, B.G., Sánchez, T.M. and Carrera, M.G. 1999. Biodiversification patterns in the Early Ordovician of Argentina. Palaios, 14: 198–214.
Received: February 15, 2003
Accepted: June 15, 2003