Major expansion of echinoderms in the early Late Ordovician (Mohawkian, middle Caradoc) and its possible causes
James SPRINKLE1 and Thomas E. GUENSBURG2
1 Department of Geological Sciences, Jackson School of Geosciences, University of Texas, Austin, TX 78712 USA. E–mail: echino@mail.utexas.edu
2 Physical Sciences Division, Rock Valley College, Rockford, IL 61114 USA
Key words: Echinoderms. Environments. Diversification. Evolution. Late Ordovician.
Introduction
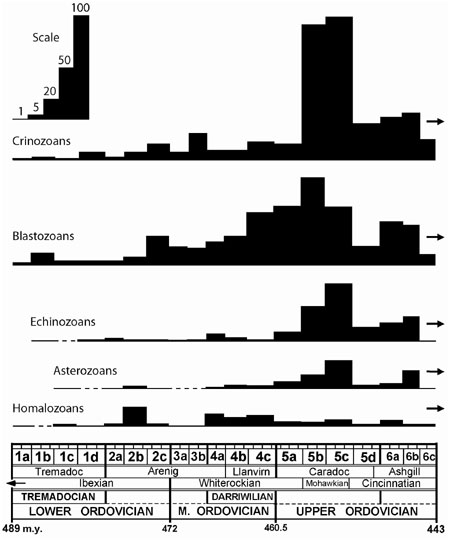
Echinoderms did not have a smooth, slowly increasing pattern of clade introduction and diversity increase in the Early Paleozoic, but a stepped, two–part radiation separated by an interval of very low diversity in the Late Cambrian. The second larger radiation in the Early to Late Ordovician ended with a huge diversity increase in the Mohawkian (middle Caradoc) that represents one of the high points of echinoderm class, clade, and species diversity for the entire Paleozoic (Sprinkle, 1980). This Mohawkian diversity spike is one of the most striking features of the early echinoderm radiation, and its possible causes are considered here. The diversity plots for echinoderm subphyla in Figure 1 are based on new species diversity diagrams for Ordovician echinoderm clades prepared for a forthcoming book chapter (Sprinkle and Guensburg, in press).
Echinoderm diversity pattern
The appearance and diversification of echinoderms during the Cambrian to produce the Cambrian Evolutionary Fauna (CEF) and during the Ordovician to produce the Paleozoic Evolutionary Fauna (PEF) set the stage for the remaining Paleozoic echinoderm record. The echinoderm part of the Cambrian Explosion was small, with only eight classes and 14 clades by the time of peak diversity in the Middle Cambrian (Sprinkle, 1980). A major decline then followed in the Late Cambrian, with only five classes and clades of echinoderms recorded. This is one of the lowest diversity levels for echinoderms during the entire Phanerozoic.

Figure 1. Species–level diversity of Ordovician echinoderm subphyla plotted on a Project 410 time scale with 19 time slices (see Webby et al., in press). Crinozoans include seven clades of arm–bearing, attached cinoids; Blastozoans include 11 clades of brachiole–bearing, mostly attached echinoderms; Echinozoans include seven clades of attached and free–living, box–like echinoderms; Asterozoans include three clades of free–living, star–shaped echinoderms; and Homalozoans include two clades of flattened, bottom–living echinoderms (see Sprinkle and Guensburg, in press).
Echinoderms underwent a second major expansion during the Ordovician Diversification as new members of the PEF added to or replaced members of the CEF. The observed echinoderm pattern agrees in general with those of other metazoan phyla, except that echinoderms appear to have produced more classes and clades than phyla such as brachiopods and bryozoans. Most of these new echinoderm clades (21 of 30) appeared in the Early Ordovician, but these new PEF clades and older reappearing CEF clades only slowly increased in diversity through the entire Early and Middle Ordovician. However, a dramatic increase in echinoderm species diversity is recorded in the Mohawkian (middle Caradoc) along with an increase to 17 classes and 29 clades, a maximum for the Ordovician. The increase in species diversity was especially dramatic in crinoids, which nearly quintupled, in echinozoans, which nearly quadrupled, and in asterozoans, which more than tripled. Blastozoans, which had been the dominant echinoderms before this time (Figure 1), show less than a 50% diversity increase, and homalozoans, which had peaked earlier in the period, remained stable (Figure 1). Crinoids then became the most diverse echinoderm class, a position they held almost continually until the end of the Paleozoic (Sprinkle, 1980). Echinoderm diversity then declined during the latest Ordovician (Figure 1) as environmental conditions gradually deteriorated, leading to the glacially driven Hirnantian mass extinction. However, 23 of the 27 late Cincinnatian or Ashgill echinoderm clades survived this extinction to reappear in the Silurian.
Intrinsic biotic factors for Mohawkian expansion
The dramatic increase in echinoderm clade and especially species diversity in the Mohawkian (middle Caradoc) probably did not have a single controlling cause, but a combination of contributing factors. One major category involves mostly intrinsic biotic features that were developed by these echinoderms during the Ordovician (Allmon and Ross, 1990). These include new ways of life for Late Ordovician echinoderms, including the first record of predation by starfish on bivalves (Blake and Guensburg, 1994), appearance of the first grazing or herbivorous echinoids (Kolata, 1975; Smith and Savill, 2002), and appearance of the flattened, circular, facet–bearing cyclocystoids that were either upright, sessile, suspension feeders (our interpretation) or inverted, mobile, deposit feeders (Smith and Paul, 1982). Lens–shaped and globular paracrinoids were another group of PEF suspension feeders that greatly diversified during the Mohawkian; they were tethered just above (or sat directly on) the sea floor (Parsley and Mintz, 1975) using a short stem and small encrusting holdfast.
There were also major improvements in morphology in existing groups of Mohawkian echinoderms, especially crinoids. Although crinoids had already developed a wide range of stem lengths, several new distal stem attachments and holdfasts were introduced by crinoids in the Middle and early Late Ordovician to attach to soft mud bottoms. These included radiating root systems to anchor a vertical stem, recumbent distal stems with long cirri draped along the sea floor (Brett, 1981), and loss of the distal holdfast to allow a drifting crinoid to cling to upright objects, such as bryozoans and other stems, by coiling its distal stem (Guensburg, 1984). Crinoids (especially camerates) also developed more advanced biserial arms with denser pinnulation for more efficient, small–particle, suspension feeding (Kolata, 1975: Guensburg, 1984). This type of small–particle feeding had previously been exploited mostly by blastozoan echinoderms using brachioles (Guensburg and Sprinkle, 1992).
Extrinsic environmental factors for Mohawkian expansion
A second major category of causes involves favorable extrinsic environmental factors (Allmon and Ross, 1990). Warm, stable, Greenhouse conditions prevailed throughout much of the Ordovician. Wide tropical and temperate climatic zones, and only small polar regions with standing ice were present (Sheehan, 2001). "Calcite Sea" conditions with rapid carbonate cementation led to many cemented grainstones and hardgrounds (Sandberg, 1983; Stanley and Hardie, 1999). High sea levels with widespread cratonic seas and wide continental shelves provided extensive living areas for echinoderms. Many of these areas had carbonates interbedded with thin shales, favorable for echinoderm preservation and recovery. Frequent storms tore loose, smothered, and rapidly buried echinoderms, resulting in many complete specimens (Regnéll, 1945; Brower and Veinus, 1974; Sprinkle, 1982; Guensburg, 1984). Many hardgrounds, shell pavements, bryozoan thickets, sponge and bryozoan mounds, and some larger reefs (Webby, 2002) provided stable hard substrates for attached echinoderms. Orogenic activity was relatively subdued in most regions, with the Taconic Orogeny only starting in mid–Mohawkian (Holland and Patskowsky, 1997). There was little delta building and limited siliciclastic input to epeiric seas on most continents. However, widespread Mohawkian bentonites were erupted from volcanic island arcs, providing useful chronostratigraphic markers (Kolata et al., 1996).
Extrinsic biogeographic factor for Mohawkian expansion
A third likely cause for high echinoderm diversity involves an extrinsic biogeographic factor, a category not discussed by Allmon and Ross (1990). Although the Iapetus Ocean had partly closed by the Mohawkian, many continents and island arcs were still isolated from each other, so echinoderm faunas were very different on many of these continents, increasing world–wide total diversity (Paul, 1976; Sprinkle and Guensburg, in press). This gradually changed during the remainder of the Late Ordovician as the Iapetus Ocean closed further and echinoderm faunas became more cosmopolitan, lowering world–wide total diversity (Paul, 1976).
Other less likely factors
Extrinsic biotic factors, such as competition, predation, and parasitism (Allmon and Ross, 1990), appear to be less likely causes for the observed Mohawkian (middle Caradoc) expansion of echinoderms. Competition among echinoderms and between echinoderms and other benthic metazoans (especially suspension feeders) increased during the Ordovician as diversity and abundance of all these groups gradually increased, but this would act as a negative factor for the diversity of any one group. The increasing competition for food and living space among benthic suspension feeders apparently did promote the evolution of longer stems and new attachment methods in crinoids and blastozoans (Ausich, 1980; Bottjer and Ausich, 1986), so it did have some ultimate if not immediate positive affect. Predation and parasitism on echinoderms remained low throughout the Ordovician, with very few specimens showing evidence of bite marks, nipped arms, or borings in the thecal plates (see rare examples in Sprinkle, 1982). Many later predators or parasites on echinoderms, such as jawed fish, shell–boring gastropods, and starfish, were either absent or had just appeared in the Ordovician.
Conclusions
1. Some combination of intrinsic biotic, extrinsic environmental, and extrinsic biogeographic factors apparently were responsible for the huge diversity increase of echinoderms in the Mohawkian (middle Caradoc) compared to other parts of the Ordovician.
2. Ordovician echinoderms had evolved several new ways of life and many new morphologic improvements for feeding and attachment that greatly increased their diversity by the Mohawkian (middle Caradoc).
3. The Mohawkian (middle Caradoc) had the highest sea levels, most shelf living area, most favorable echinoderm living conditions, and greatest preservation potential of any time interval during the Ordovician, thus increasing echinoderm diversity.
4. Although the Iapetus Ocean had partly closed by the Mohawkian (middle Caradoc), many continents and island arcs were still isolated from each other, so echinoderm faunas were still very different on these living areas, leading to high world–wide total diversity.
5. Extrinsic biotic factors (competition, predation, and parasitism) appear to be less likely causes for the observed Mohawkian (middle Caradoc) expansion of echinoderm diversity.
Acknowledgements
This paper is a contribution to IGCP Project 410, The Great Ordovician Biodiversification Event. Field work for this project was funded by NSF Grants BSR–8906568 (Sprinkle) and EAR–9304253 (Sprinkle, Guensburg), by CRDF Cooperative Research Grant RG1–242 (Sprinkle, Guensburg, Rozhnov), and by a PRI–ACS Grant to Mark Wilson, College of Wooster (Guensburg). The Geology Foundation, University of Texas, Austin, and Rock Valley College, Rockford, IL, provided funds for travel and manuscript preparation. Chris Schneider, University of Texas, Austin, prepared Figure 1. William I. Ausich, Ohio State University, and Guillermo F. Aceñolaza, National University of Tucumán, reviewed the completed manuscript and made helpful suggestions.
References
Allmon, W.D. and Ross, R.M. 1990. Specifying causal factors in evolution: The paleontological contribution. In: Ross, R.M. and Allmon, W.D. (Eds.), Causes of Evolution: A Paleontological Perspective, University of Chicago Press, Chicago: 1–17.
Ausich, W.I. 1980. A model for niche differentiation in Lower Mississippian crinoid communities. Journal of Paleontology, 54(2): 273–288.
Blake, D.B. and Guensburg, T.E. 1994. Predation by the Ordovician asteroid Promopalaester on a pelecypod. Lethaia, 27(3): 235–239.
Bottjer, D.J. and Ausich, W.I. 1986. Phanerozoic development of tiering in soft substrata suspension–feeding communities. Paleobiology, 12(4): 400–420.
Brett, C.E. 1981. Terminology and functional morphology of attachment structures in pelmatozoan echinoderms. Lethaia, 14(4): 343–370.
Brower, J.C. and Veinus, J. 1974. Middle Ordovician crinoids from southwestern Virginia and eastern Tennessee. Bulletins of American Paleontology, 66(283): 1–125.
Guensburg, T.E. 1984. Echinodermata of the Middle Ordovician Lebanon Limestone, central Tennessee. Bulletins of American Paleontology, 86(319): 1–100.
Guensburg, T.E. and Sprinkle, J. 1992. Rise of echinoderms in the Paleozoic Evolutionary Fauna: significance of paleoenvironmental controls. Geology, 20(5): 407–410.
Holland, S.M. and Patzkowsky, M.E. 1997. Distal orogenic effects on peripheral bulge sedimentation: Middle and Upper Ordovician of the Nashville Dome. Journal of Sedimentary Research, 67(2): 250–263.
Kolata, D.R. 1975. Middle Ordovician echinoderms from northern Illinois and southern Wisconsin. Paleontological Society Memoir, 7: 1–74.
Kolata, D.R., Huff, W.D., and Bergström, S.M. 1996. Ordovician K–bentonites of eastern North America. Geological Society of America Special Paper, 313: 1–84.
Parsley, R.L. and Mintz, L.W. 1975. North American Paracrinoidea: (Ordovician: Paracrinozoa, new, Echinodermata). Bulletins of American Paleontology, 68(288): 1–115.
Paul, C.R.C. 1976. Palaeogeography of primitive echinoderms in the Ordovician. In: Bassett, M.G. (ed.), The Ordovician System, University of Wales Press and National Museum of Wales, Cardiff: 553–574.
Regnéll, G. 1945. Non–crinoid Pelmatozoa from the Paleozoic of Sweden. Meddelanden fran Lunds Geologisk–Mineralogiska Institution, 108: 1–255.
Sandberg, P.A. 1983. An oscillating trend in Phanerozoic carbonate mineralogy. Nature, 305: 19–22.
Sheehan, P.M. 2001. The Late Ordovician mass extinction. Annual Review of Earth and Planetary Sciences, 29: 331–364.
Smith, A.B. and Paul, C.R.C. 1982. Revision of the class Cyclocystoidea (Echinodermata). Philosophical Transactions, Royal Society of London, B. Biological Sciences, 296(1083): 577–684.
Smith, A.B. and Savill, J.J. 2002 (2001). Bromidechinus, a new Middle Ordovician Echinozoa (Echinodermata), and its bearing on the early history of echinoids. Transactions of the Royal Society of Edinburgh: Earth Sciences, 92(2): 137–147.
Sprinkle, J. 1980. An overview of the fossil record. In: Broadhead, T.W. and Waters, J.A. (Eds.), Echinoderms, Notes for a Short Course, University of Tennessee Department of Geological Sciences, Studies in Geology Nº 3: 15–26.
Sprinkle, J. (Ed.). 1982. Echinoderm Faunas from the Bromide Formation (Middle Ordovician) of Oklahoma. University of Kansas Paleontological Contributions, Monograph 1: 1–369.
Sprinkle, J. and Guensburg, T.E. In press. Crinozoan, blastozoan, echinozoan, asterozoan, and homalozoan echinoderms. In: Webby, B.D., Droser, M.L., Paris, F., and Percival, I (Eds.), The Great Ordovician Biodiversification Event, Columbia University Press, New York.
Stanley, S.M. and Hardie, L.A. 1999. Hypercalcification: Paleontology links plate tectonics and geochemistry to sedimentology. GSA Today, 9(2): 1–7.
Webby, B.D. 2002. Patterns of Ordovician reef development. In: Kiessling, W., Flugel, E., and Golonka, J. (Eds.), Phanerozoic Reef Patterns. SEPM Special Publication Nº 72: 129–180.
Webby, B.D., Cooper, R.A., Bergström, S.M., and Paris, F. In press. Stratigraphic framework and time slices. In: Webby, B.D., Droser, M.L., Paris, F., and Percival, I (Eds.), The Great Ordovician Biodiversification Event, Columbia University Press, New York.
Received: February 15, 2003
Accepted: June 15, 2003